Review 3D printing material considerations for medical prototypes, anatomical models, surgical guides, fixtures and application-specific engineering validation.

Medical Requirements for Printed Materials
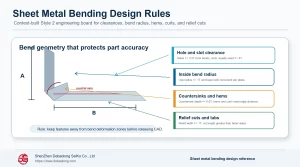
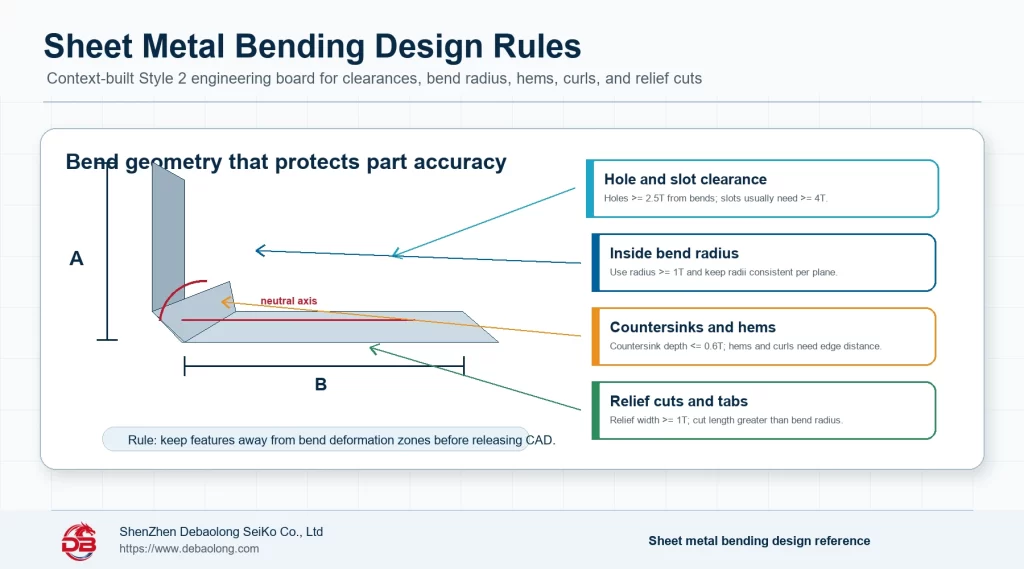
Medical-related 3D printing materials must be evaluated by use case. A training model, anatomical model, surgical planning aid, lab fixture and functional medical component all have different requirements for accuracy, cleaning, strength, documentation and contact risk.
Material selection should not be made from generic strength alone.
In practical terms, medical requirements for printed materials should be translated into drawing notes, process limits, material choices, tolerance expectations, finish requirements and inspection points before the design is released. For 3d printing materials for medical applications: engineering selection guide, the useful result is not just a general comparison, but a manufacturing decision that buyers, engineers and suppliers can apply consistently during quoting, prototyping and production planning. This is where a topic moves from educational guidance into a real release standard.
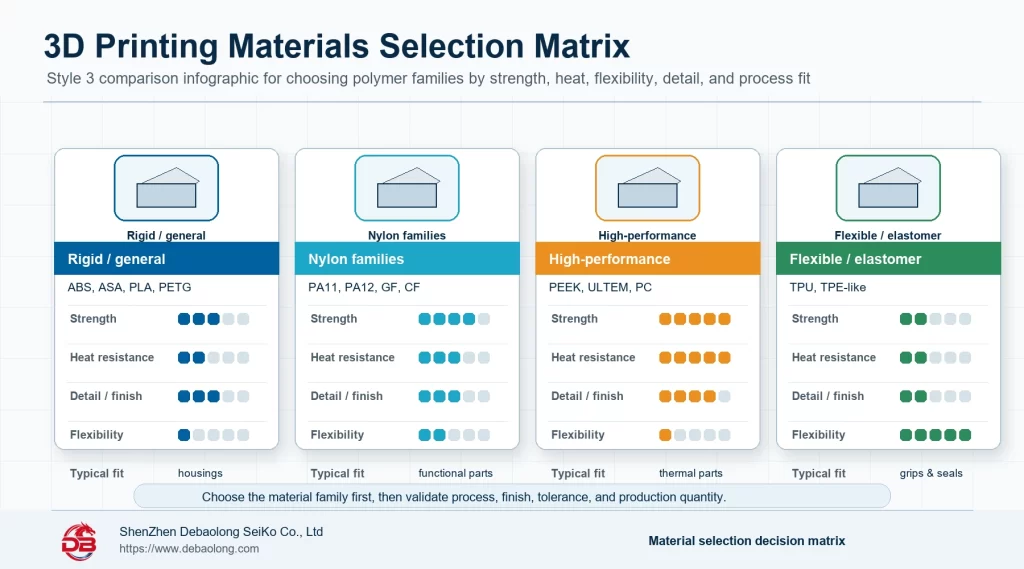
PA12 Nylon, ULTEM, Silicone-Like Materials and Metals
The source article covers material families such as PA12 nylon, ULTEM 1010, silicone-like materials, titanium Ti6Al4V and stainless steel 17-4PH. Nylon can support durable functional parts, ULTEM can support higher temperature needs, silicone-like materials can support soft-touch models, and metals require stronger qualification and post-processing.
Each material should be matched with process capability and documentation needs.
In practical terms, pa12 nylon, ultem, silicone-like materials and metals should be translated into drawing notes, process limits, material choices, tolerance expectations, finish requirements and inspection points before the design is released. For 3d printing materials for medical applications: engineering selection guide, the useful result is not just a general comparison, but a manufacturing decision that buyers, engineers and suppliers can apply consistently during quoting, prototyping and production planning. This is where a topic moves from educational guidance into a real release standard.
Accuracy, Sterilization and Post-Processing
Medical-related parts may require dimensional checks, cleaning, curing, surface finishing or sterilization compatibility. These steps can affect material behavior and part dimensions.
Assumptions are risky: the selected material and post-process must match the intended application.
In practical terms, accuracy, sterilization and post-processing should be translated into drawing notes, process limits, material choices, tolerance expectations, finish requirements and inspection points before the design is released. For 3d printing materials for medical applications: engineering selection guide, the useful result is not just a general comparison, but a manufacturing decision that buyers, engineers and suppliers can apply consistently during quoting, prototyping and production planning. This is where a topic moves from educational guidance into a real release standard.
Responsible Use in Medical Development
3D printing can accelerate medical device development, but printed prototypes should have clear boundaries. They may prove fit, visualization or handling while final validation requires controlled manufacturing and additional testing.
DEBAOLONG connects printed prototypes with downstream precision manufacturing, inspection and documentation needs.
In practical terms, responsible use in medical development should be translated into drawing notes, process limits, material choices, tolerance expectations, finish requirements and inspection points before the design is released. For 3d printing materials for medical applications: engineering selection guide, the useful result is not just a general comparison, but a manufacturing decision that buyers, engineers and suppliers can apply consistently during quoting, prototyping and production planning. This is where a topic moves from educational guidance into a real release standard.
Choosing Materials for Real Medical Development
The source article covers several medical-printing material families, but the real decision depends on use case. Visual anatomical aids, handling models, lab fixtures and functional medical components each require different levels of process control, cleaning, documentation and post-processing discipline.
DEBAOLONG uses that distinction to decide whether a printed medical part should remain a development tool or move into a more controlled manufacturing plan. This prevents prototype convenience from being mistaken for final-use suitability when sterility, dimensional control or clinical environment requirements are significant.
In practical terms, choosing materials for real medical development should be translated into drawing notes, process limits, material choices, tolerance expectations, finish requirements and inspection points before the design is released. For 3d printing materials for medical applications: engineering selection guide, the useful result is not just a general comparison, but a manufacturing decision that buyers, engineers and suppliers can apply consistently during quoting, prototyping and production planning. This is where a topic moves from educational guidance into a real release standard.
DEBAOLONG Engineering Review
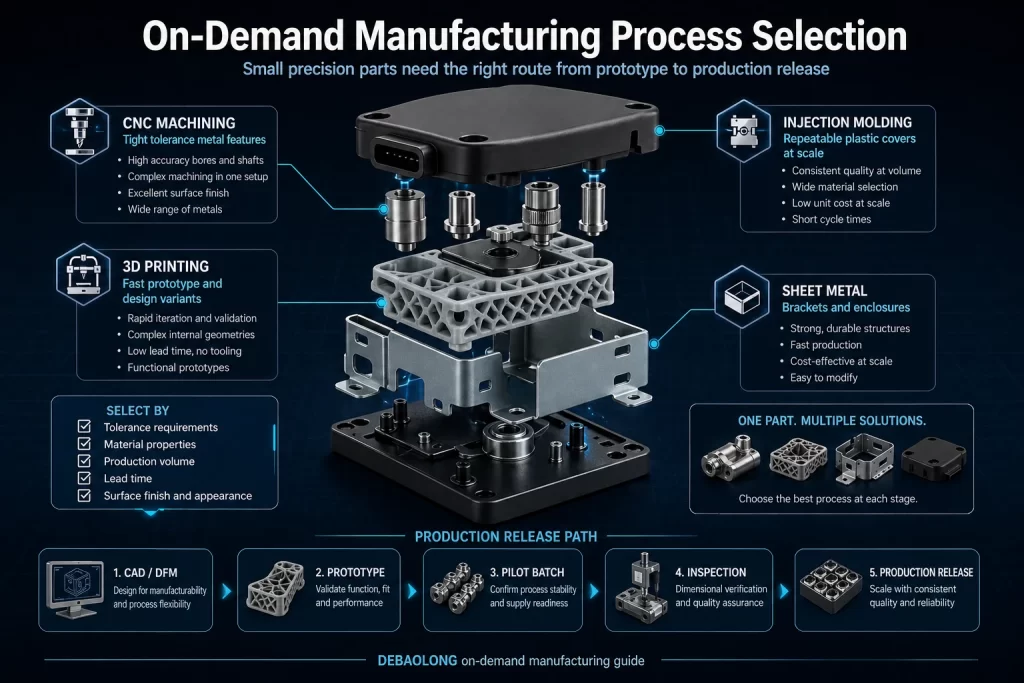
This article has been rebuilt around the source article’s actual engineering logic instead of being treated as a short summary. The purpose is to preserve the useful decision sequence: process capability, material fit, tolerance impact, finish expectations, production risk, validation steps and how the next manufacturing decision should be made.
Before release, the design team should confirm the intended application, required quantity, critical dimensions, operating environment, acceptable finish, cleaning requirements where relevant, and the inspection method that will be used to validate the part. These details turn a manufacturing article into a production-ready review standard and reduce late changes after prototyping or tooling work has already started.
For broader manufacturing planning, review the DEBAOLONG Manufacturing Engineering Knowledge Center, compare major manufacturing process options, or use DFM for prototyping to connect early design decisions with production-ready parts.
FAQ
How should engineers use this 3d printing materials for medical applications: engineering selection guide guide?
Use it as a practical release checklist before quoting, prototyping or production handoff. The best result is a clearer manufacturing decision, not simply a longer list of process facts.
Why does source-aligned structure matter in this topic?
Because each source section answers a different engineering question. Keeping those questions separate makes it easier to review geometry, material, cost, finish, quantity and production risk without silently collapsing useful detail.
Can DEBAOLONG help turn the review into a manufacturable plan?
Yes. DEBAOLONG can review geometry, materials, tolerances, finish requirements, visual expectations and production intent before the project moves into manufacturing.